 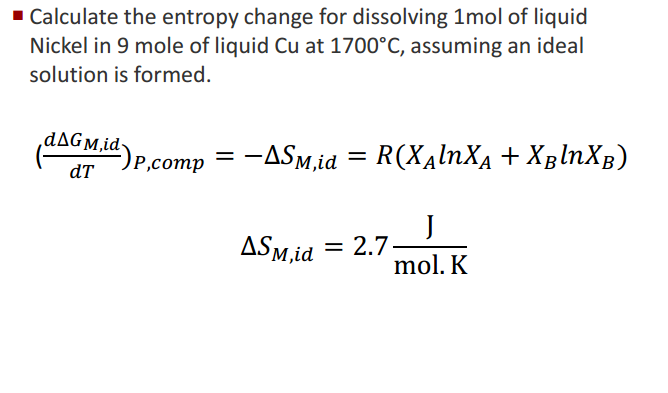
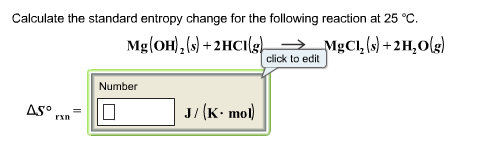
That means that if you are calculating entropy change, you must multiply the enthalpy change value by 1000. 
But will serve as a decent guideline for guessing what the entropy should be. But entropy change is quoted in energy units of J. This type of rational does not always work (think of a scenario with hundreds of outcomes all dominated by one occurring \(99.999\%\) of the time). We can redefine entropy as the expected number of bits one needs to communicate any result from a distribution. The two formulas highly resemble one another, the primary difference between the two is \(x\) vs \(\log_2p(x)\). If instead I used a coin for which both sides were tails you could predict the outcome correctly \(100\%\) of the time.Įntropy helps us quantify how uncertain we are of an outcome. BYJU’S online entropy calculator tool makes the calculation faster, and it displays the entropy in a fraction of seconds. #Entropy change calculator free#For example if I asked you to predict the outcome of a regular fair coin, you have a \(50\%\) chance of being correct. Entropy Calculator is a free online tool that displays the entropy change for the chemical reaction. So, if we want to calculate the total entropy change of these systems, we need to calculate the entropy change of the water over the different periods. The vibrational temperature of 'H'2 (the temperature at which it accesses its vibrational states) is Theta(vib) '6333 K'. #Entropy change calculator how to#The higher the entropy the more unpredictable the outcome is. How to calculate the change in the entropy of hydrogen molecule before and after the adsorption Chemistry Thermochemistry Entropy. Essentially how uncertain are we of the value drawn from some distribution. You just have to work out the expression for dS and integrate from T1 to T2 to get ΔS body.Quantifying Randomness: Entropy, Information Gain and Decision Trees EntropyĮntropy is a measure of expected “surprise”. Each step would result in a heat flow dQ = mCdT. #Entropy change calculator series#A reversible process for this would be to successively put it in contact in series with an arbitrarily large number of heat baths - each of which are at an infinitesimally higher temperature than the body - until it reaches T2. The solid body changes temperature from T1 to T2. The reversible path would simply be a reversible isothermal heat flow out of the heat bath of Q, where Q is the amount of heat flow into the solid body. Here you are assuming that the bath has an arbitrarily large heat capacity so its temperature before and after the process is the same. Since the actual process was NOT reversible, the reversible paths will not be the same path for each component. You just have to find a reversible path for each component (ie the solid body and the bath). the direction of heat flow) by making an infinitesimal change in conditions.īut to find the change in entropy of each body, you only look at the initial and final states - you do not care about the process that occurred in going from the initial to final states. The process is not reversible because you cannot change the direction of the process (ie. I try to solve this problem by constructing 1 isotherm and 1 adiabat to replace original isochoric path and find out that dS actually = dQ / T = Cv/T dT ,Īlthough i know where this come from ( dS = dQ /T) from maths wayīut I think i misunderstand some physical conceptĬan someone explain why dS = dQ/T, even when this process is not reversible in some physical way? There comes my question, we find out that this isochoric process is irreversibleīut when we use the equation dS = dQ/T, we could only use it in reversible process. So total entropy is not zero when T2 and T1 has a finite change. This free online devise entropy calculator is helpful to find the change in entropy for a reaction, change in Gibbs free energy and change in entropy of. 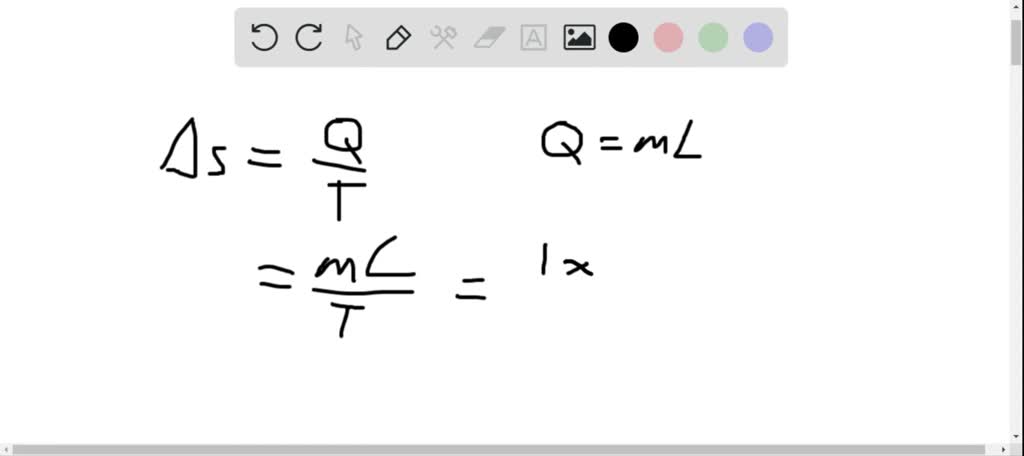
Then i compute the entropy change of heat body in isochoric system = nR ln(T2/T1)Īnd entropy change of heat bath = - Cv(T2-T1)/T2 ( i read these from notes actually but i am confused with the reversibility in it)īut I remember that we have proved beforeĭS >= dQ/T for all process, and equality hold when process is reversible(like isotherm and adiabats) We could calculate its entropy change(heat body) from dS = Cv/T dTĪnd entropy change of heat bath from dS = dQ / T2, using dQ = Cv dT in 1st equation In an isochoric process ( a solid heat body with T1 temp. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
